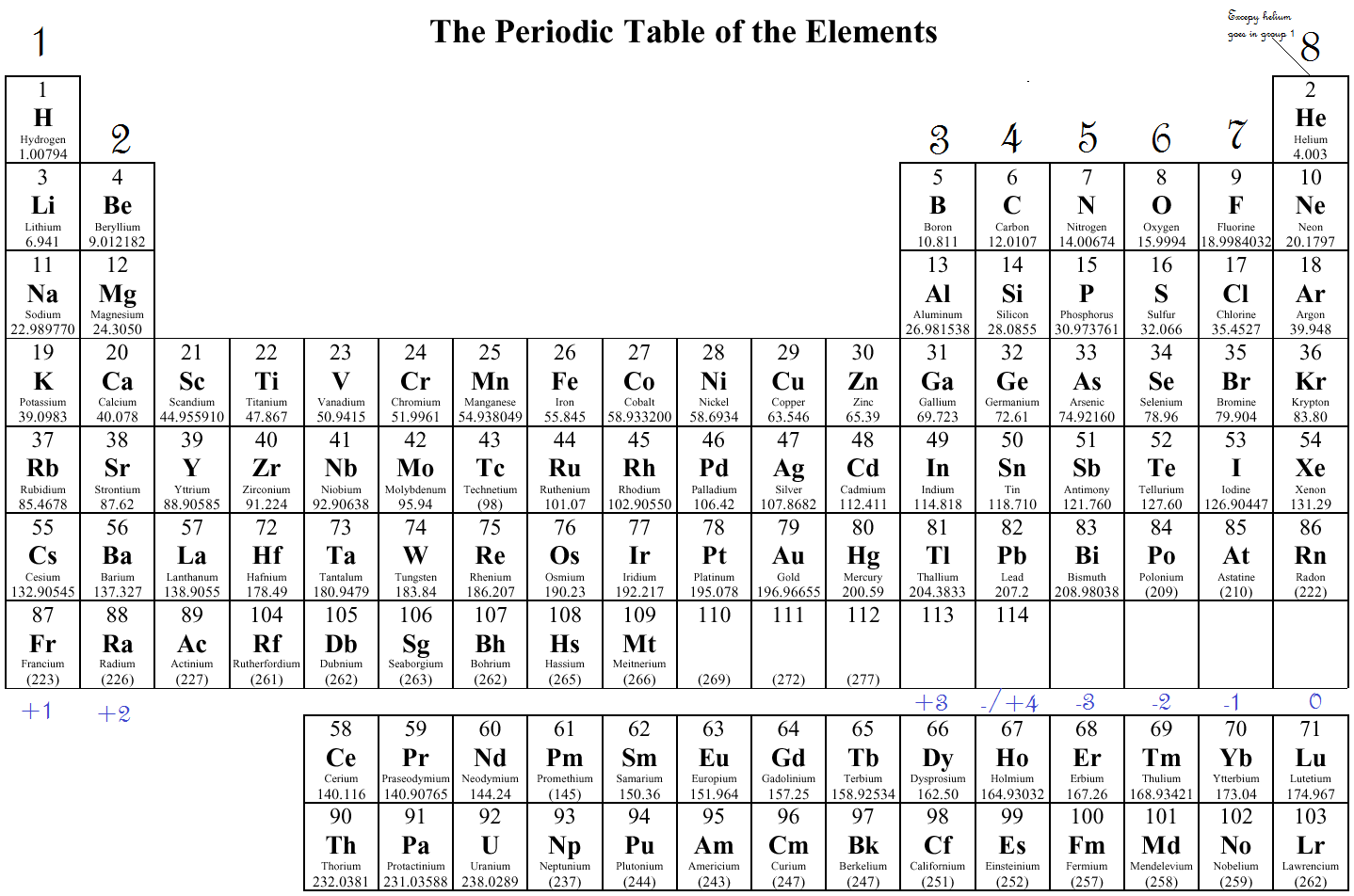
This is because of the stability of the valence shell.

Each electron in an atom has an associated ionization energy that can be measured. Conceptually, this is the exact opposite of electron affinity. This is the force required to remove an electron from an atom when it is gaseous, or the propensity of an atom to lose electrons. This is because their inability to attract electrons as readily as other elements is impacted by their metallic characteristics. Although the transition metals have electronegativity values, there is little variation in their values across periods and within groups.As a result, the electronegativity values of noble gases, lanthanides, and actinides are zero. The chemistry of the lanthanides and actinides is more complex and doesn’t typically follow any patterns. The noble gases have a full valence shell and often do not draw electrons. The noble gases, lanthanides, and actinides are significant exceptions to the aforementioned trend.
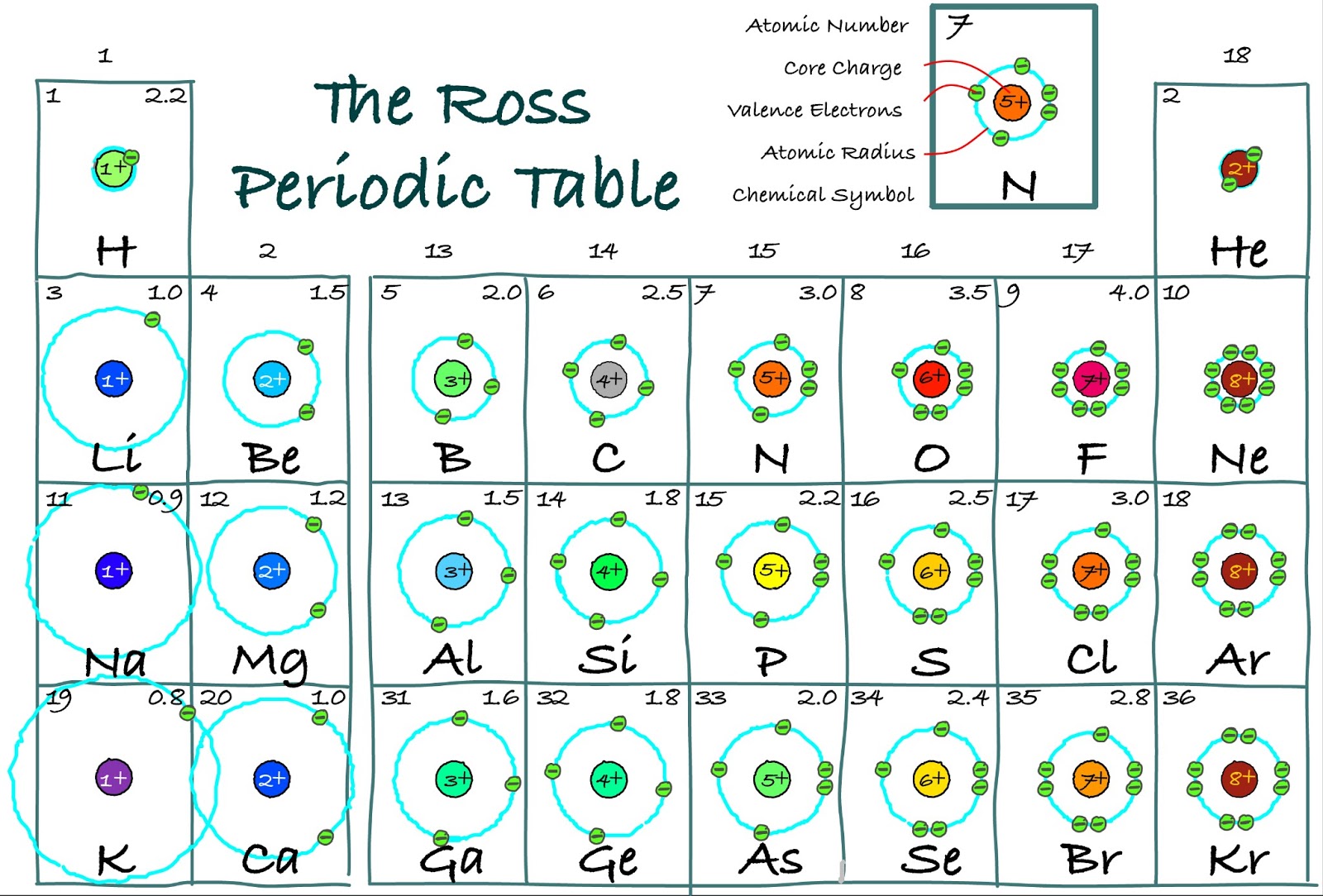
The electronegativity of a group decreases from top to bottom: This is due to the fact that as atomic number decreases within a group, the atomic radius-the separation between the valence electrons and nucleus-increases.Electronegativity rises from left to right across a period: It is simpler to remove an electron into the valence shell than to contribute one if it is more than halfway full.This property reflects how easily an atom can form a chemical bond. Electronegativity is a measure of attraction an atom has for bonding electrons.
